Description
Product Overview
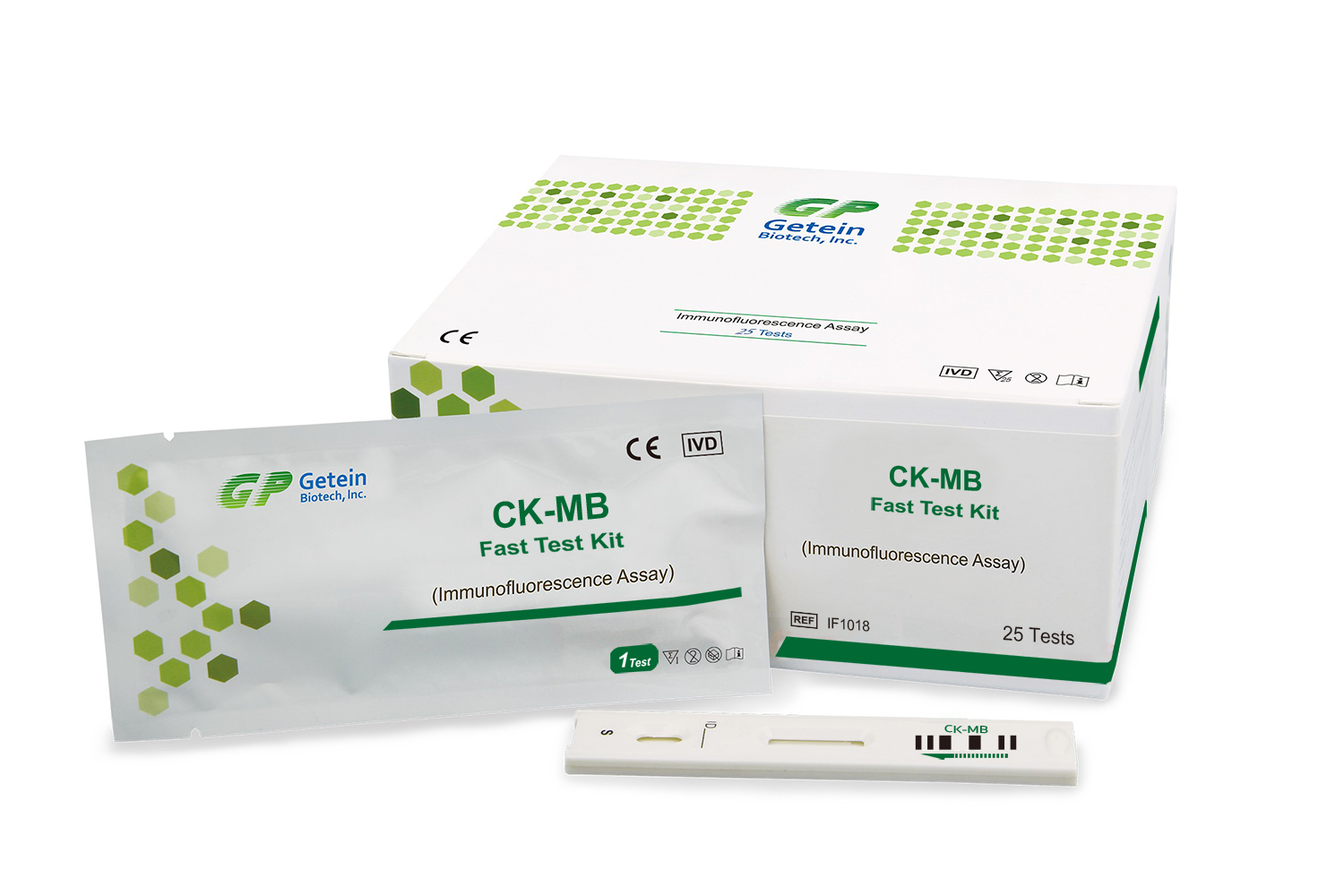
The CK-MB Test Kit is designed for quantitative determination of creatine kinase-MB (CK-MB) in human whole blood, serum or plasma using fluorescence immunoassay technology. CK-MB is an isoenzyme of creatine kinase that is primarily found in cardiac muscle cells.
Measurement of CK-MB levels assists clinicians in diagnosing myocardial infarction and evaluating cardiac muscle injury in patients presenting with chest pain.
Clinical Significance
CK-MB is released into the bloodstream when cardiac muscle cells are damaged. Elevated CK-MB levels are associated with myocardial infarction and other cardiac injuries.
Measurement of CK-MB is useful for:
• Diagnosis of acute myocardial infarction
• Monitoring cardiac muscle injury
• Evaluation of chest pain in emergency departments
• Monitoring recovery after cardiac events
Key Features
• Quantitative fluorescence immunoassay
• Rapid cardiac marker detection
• High sensitivity measurement
• Suitable for hospital laboratories and emergency departments
• Compatible with immunofluorescence analyzers
Sample Type
Whole blood
Serum
Plasma
Test Time
Approximately 15 minutes
Compatible Analyzers
Compatible with fluorescence immunoassay analyzers including GETEIN 1100, GETEIN 1160 and FIA 8000 analyzers.
Principle of the Test
The CK-MB Test Kit is based on fluorescence immunoassay technology. The sample reacts with fluorescence labeled antibodies specific to CK-MB. The fluorescence signal generated is measured by the immunofluorescence analyzer and is proportional to the concentration of CK-MB present in the sample.
Applications
This product can be effectively utilized in:
• Emergency departments
• Hospital laboratories
• Cardiac care units
• Point-of-care testing centers
Intended Use
The CK-MB Test Kit is intended for in vitro diagnostic use for quantitative determination of CK-MB in human whole blood, serum or plasma using fluorescence immunoassay analyzers in clinical laboratories and hospital settings.
Related Tests
Related cardiac marker tests include Troponin I Test Kit, NT-proBNP Test Kit and D-Dimer Test Kit used in hospital emergency diagnostics.